BI, Testpack, and PCD Sterilization Supplies
Customers may purchase BIs, Testpacks and PCD sterilization supplies from Nelson Labs for validation and ongoing process monitoring of the validated sterilization cycle.
Biological Indicators (BIs)
Biological indicators are an essential sterilization process monitor with a known population of microorganisms on the spore strip, typically 106 for sterilization monitors, and with the most resistant organism (MRO) for the sterilization process.
Process Challenge Devices and Testpacks (PCD/TP)
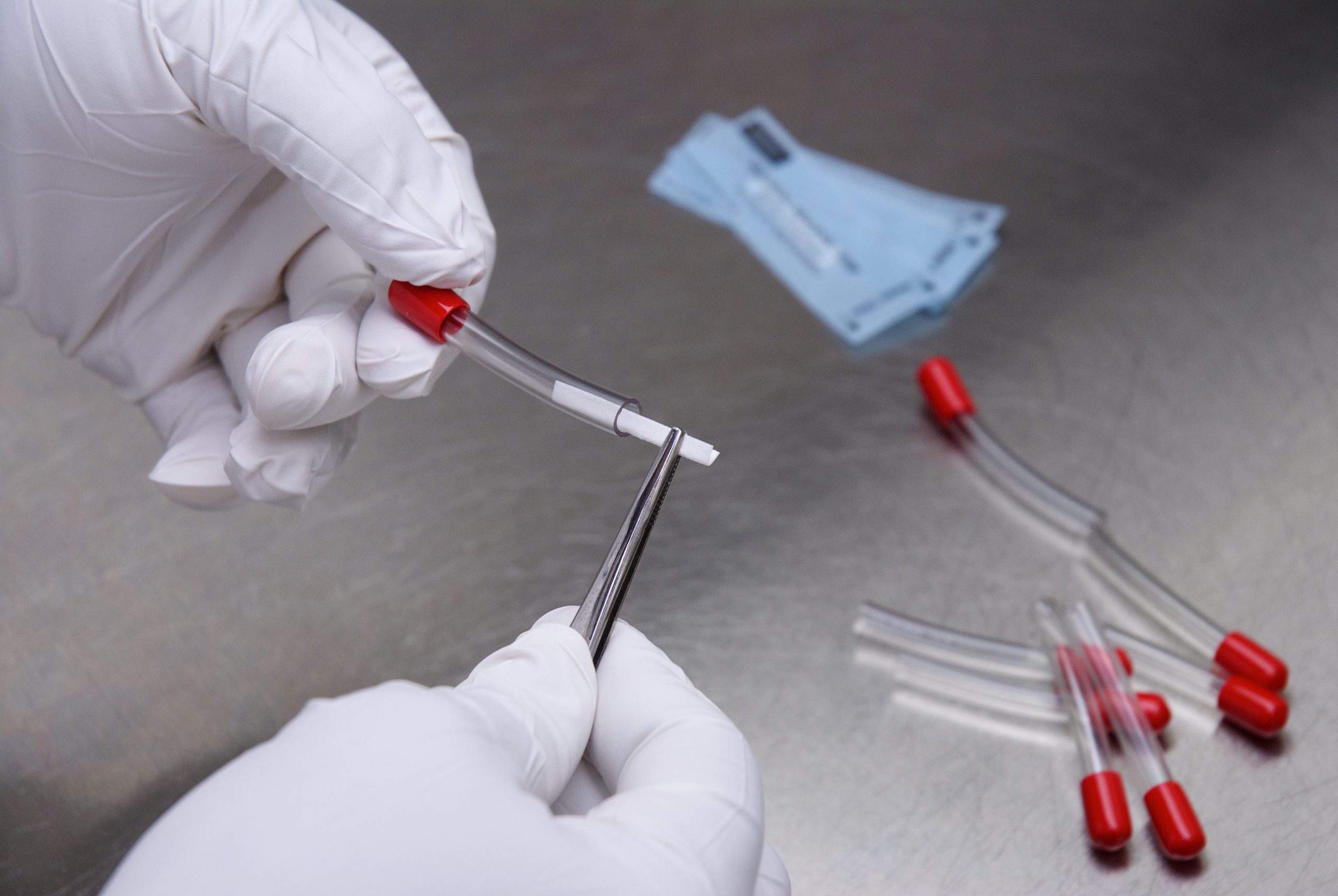
Process Challenge Devices and Testpacks serve to provide biological monitors for terminal sterilization of single-use devices processed in a manufacturing setting. These products are not intended to monitor sterilization processes used in health care facilities.
PCDs and Testpacks are surrogates of product used for microbiological monitoring of both validation and routine sterilization cycles. It consists of a biological indicator within a specific configuration such as syringes, tubing, etc., that poses an increased resistance to represent a stringent challenge to the sterilization process.
Nelson Labs produces different configurations of PCDs and Testpacks designed with consideration to the various sterilization parameters. Their composition is based on different resistance levels to adapt to different cycle specifications.
To Place An Order
- Download PCD Supply Order Form
- Download How to Order EO Sterilization Supplies
- Download PCD/TP Specification Sheet
Applicable Standards
- ISO 11135
- ISO 14161