
Applicable Standards
- ISO 10993
- ISO 18562
- US FDA Guidance Document on ISO 10993-1 (Sep 2016)
- MDR Regulation (EU) 2017/745 of the European Parliament and the Council of 05 April 2017 on Medical Devices
The days of the “check box” approach to biocompatibility are over.
Instead manufacturers of modern-day, complex devices need to assess the biocompatibility of their medical device materials and processes by taking a holistic risk-based approach to their biological safety evaluations.
Our experts have broken out the biological safety evaluation process into three distinct phases that can be used to accommodate a medical device in any stage of the product life-cycle.
This process is comprehensive and available today:
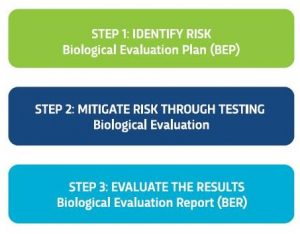
Biological Evaluation Plan (BEP)
A Biological Evaluation Plan (BEP) is an initial risk assessment offered by Nelson Labs is intended to meet the objectives outline in ISO 10993-1, clause 4 “General principles applying to biological evaluation of medical devices.” The first part of this clause states specifically that “The biological evaluation shall be planned, carried out, and documented by knowledgeable and experienced professionals.” The BEP takes into consideration the materials, processing, and historical use of the device. The deliverable is a signed biological evaluation plan recommending testing and information needed to demonstrate device safety.
Biological Evaluation (Testing) and Risk Assessments
Carry out the testing outlined in the BEP. This testing can include chemistry testing, biocompatibility testing, and/or written risk assessments.
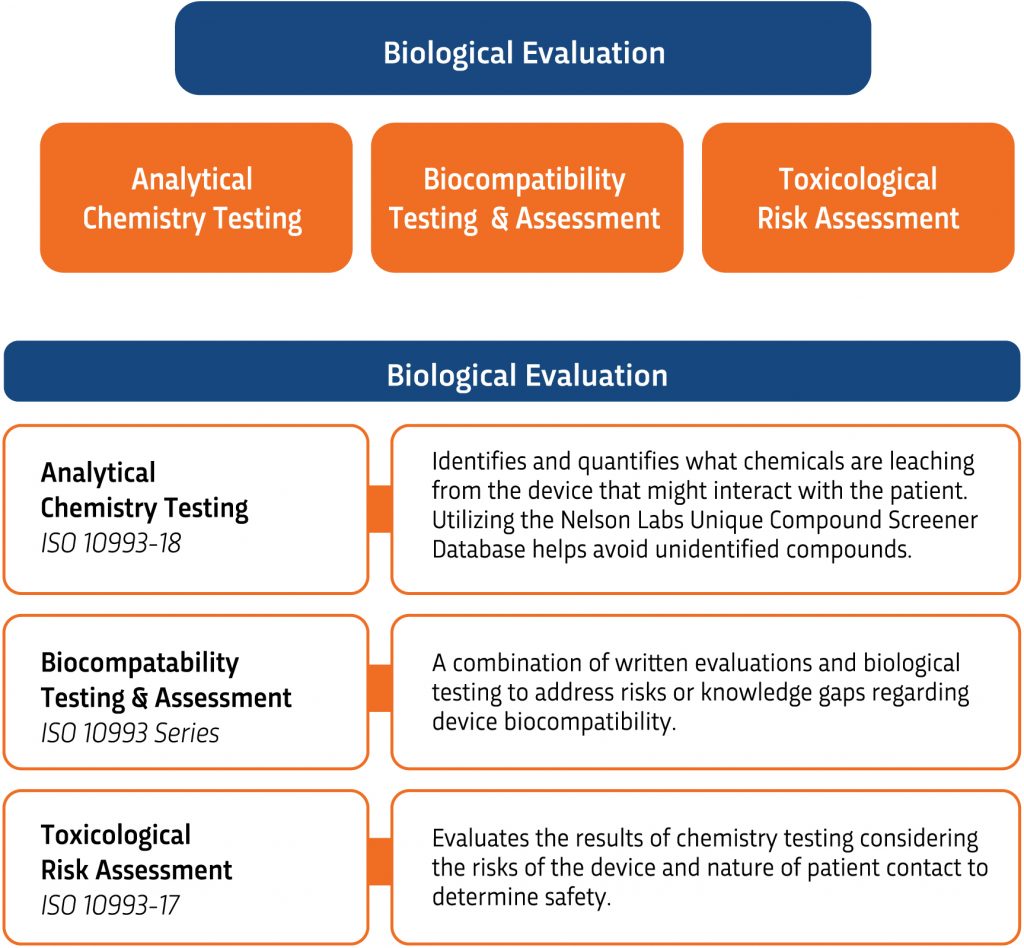
A Toxicological Risk Assessment (TRA) assesses patient risks that may arise from exposure to chemical constituents extracted, leached, or otherwise released from a medical device during clinical use. The applicable guidelines followed may depend on the route of exposure and the type of data assessed. The following are examples:
- Extractable substances released from a medical device are assessed following ISO 10993-17:2023.
- Volatile compounds released from breathing gas pathway devices are assessed per ISO 18562:2024.
- Extractables and leachables in drug-device combination products are assessed per ISO 10993-17, ICH M7, and ICH Q3-series.
- A materials-based TRA is used to assess exposure to formulation ingredients that are ingested or absorbed.
Nelson Labs toxicologists also prepare customized technical opinion memos to provide defensible scientific justification of applicable test conditions, findings, or chemical-specific toxicological profiles. The following are examples:
- Provide rationale to support the validity of extraction conditions (solvents matching the polarity of biological fluids or tissues, clinically relevant temperatures, duration of extraction, or flow rate conditions).
- Provide rationale supporting negligible toxicological risks based on manufacturing changes or nonconformances.
- Evaluate compliance to chemical-specific regulatory requirements (California Prop. 65 or EU CMR).
Biological Evaluation Report (BER)
The Biological Evaluation Report (BER) is designed to meet the requirements outline in ISO 10993-1:2018, clause 7: “Expert assessors who have the necessary knowledge and experience shall determine and document:
- the strategy and planned content for the biological evaluation of the medical device;
- the criteria for determining the acceptability of the material for the intended purpose, in line with the risk management plan;
- the adequacy of the material characterization;
- the rationale for selection and/or waiving of tests;
- the interpretation of existing data and results of testing;
- the need for any additional data to complete the biological evaluation;
- overall biological safety conclusions for the medical device.”
Services Summary
Nelson Labs offers a full trifecta of services to meet the requirements of ISO 10993 for Extractables & Leachables, Biocompatibility and Toxicological Risk Assessments of medical devices. Biocompatibility testing is very common in the medical device industry. However, with 24 possible categories, each with a unique set of testing requirements, the biocompatibility testing experience can be intimidating. Even after a device has been categorized, a myriad of decisions for each test remains.
Our Expert Advisory Services group can provide a full service experience. If you are familiar with ISO 10993 and its requirements, you may also select services you need below. Every medical device must be subjected to three common biocompatibility tests (often referred to as the Big Three): cytotoxicity, sensitization, and irritation tests. Each test screens for the presence of toxic, leachable materials. Based on the category of the device, as many as five additional testing categories may be required (see test matrix).
If you have additional questions about Biocompatibility & Toxicology testing, or would like to consult with the experts at Nelson Labs, just send us a request or call us at +1 (801) 290-7500.


